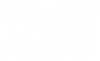
TAKE CARE OF YOUR CLINICAL PROJECT
WITH EXPERT SUPPORT
WITH EXPERT SUPPORT
OUR NEWS
300+
COMPLETED
STUDIES
100+
PUBLICATIONS
4000+
CITATIONS
OUR SPECIALIZATIONS
A CRO with over 20 years’ experience, compliant with standards (ICH, CDISC, etc.) and regulatory requirements (ANSM, EMA, FDA…).
Working with life sciences stakeholders, we design, set-up, and coordinate studies throughout Europe thanks to our network, from the writing of protocols to the publication of scientific articles.
PHARMA LABS – BIO/MEDTECHs – MD/IVD – COSMETICS – NUTRITION – VETERINARY

Interventional research
Phase II to IV studies
All designs

Interventional research
with minimal risk/constraint
with minimal risk/constraint
Transversal or longitudinal studies
All designs

Non-interventional research
Cohort studies, registries
Observatories, Post-MA studies

Historical data studies
Bibliographic studies
Meta-analyses
OUR SERVICES
CASE STUDIES
-
Hospital - Hosp(EN)
Hospital
Hospital
Develop and demonstrate the reliability of an automated measurement system for a diagnostic evaluation test for a disease.
-
Biotech - Biotech (EN)
Biotech
Biotech
Determine the dose-response relationship and dosage to be used during a subsequent study phase for comparing the drug to placebo.
-
Diagnostic - Diag (EN)
Diagnostic
Diagnostic
Develop a clinical evaluation strategy for the CE marking of an image-processing algorithm.
-
Pharma - Pharma (EN)
Pharma
Pharma
Set up and lead a prospective, multicenter phase IV clinical trial.
-
Nutrition - Nutrition (EN)
Nutrition
Nutrition
Develop and apply a statistical (propensity score) and medical analysis method for a non-randomized study.
-
Medical Device - Meddev (EN)
Medical Device
Medical device
Set up and lead a PMCF (post marketing clinical follow-up) and patient recurrence evaluation study.
- Hospital
- Biotech
- Diagnostic
- Pharma
- Nutrition
- Medical Device
OUR REFERENCES
Soladis Clinical Studies - France
15 boulevard du Général Leclerc
59100 Roubaix - FRANCE
Tél: +33(0)3.28.09.94.70
Soladis - France (HQ)
6-8 rue Bellecombe
69006 LYON - FRANCE
Tél: +33(0)4.72.83.86.70
Soladis GmbH - Switzerland
Lange Gasse 15
CH-4052 Basel
Phone: +41(0)58.258.15.80
Soladis Inc. - USA
185 Alewife Brook Pkwy, Unit 210
Cambridge, MA 02138-1100
Phone: (+1)857.675.1189
Contact us
SEARCH


























